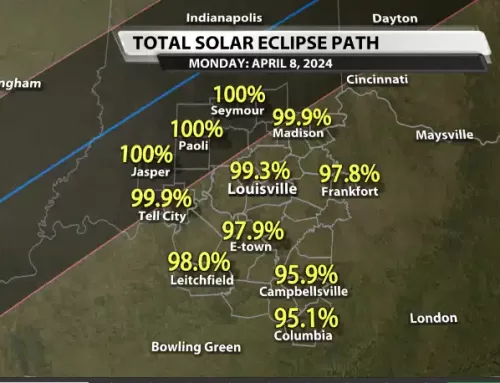
By Eli Hughes–
The University of Louisville received $8.5 million in December for funding the development and testing of a nasal spray using a compound that has shown success in inhibiting the virus that causes COVID-19, also known as SARS-CoV-2. The funding will be provided by the Department of Defense and will allow researchers to create the nasal spray and complete the phase one clinical trial.
The project makes use of a protein known as Q-Griffithsin, which was developed and co-owned by U of L. The protein was originally discover in red algae, but it can be easily replicated in tobacco plants, which are more accessible in Kentucky. The protein was initially being looked at as a treatment for HIV but had shown activity against past coronaviruses.
“HIV has been the big focus in the development of the project so far, and we have a clinical trial on going for that aspect. But when CoV-2 started we began looking at if we would have activity against CoV-2 as well,” said Joshua Fuqua, an instructor and researcher for U of L’s Department of Pharmacology and Toxicology who will be managing the project.
“Because we had activity against other coronaviruses in the past and the regional biocontainment lab had access to SARS-CoV-2. So we gave them some of that drug and they were able to test it relatively quickly and give us some understanding that it had activity [against SARS-CoV-2.]”
The product they are developing is designed to be taken daily or every other day through the nose to help prevent contracting COVID-19. The treatment would act as a protective coating in the sinuses, which is where the body first comes in contact with the virus. The protein would then inhibit the virus and keep it from multiplying and infecting the rest of the body.
Even though the Phizer and Moderna vaccines are now available, this treatment can still offer unique benefits, such as broader activity than the vaccines and the potential to help individuals who can’t get the vaccine.
“If you have viral mutations, or things like that, we are most likely going to have activity still. A virus can mutate where a vaccine is no longer effective, but we can have broader activity,” Fuqua said. He believes that the vaccines that are coming out now are great vaccines and will be useful in fighting this virus, but they are also very specific to SARS-CoV-2, in a way that this treatment might not be since it has shown activity in past coronaviruses.
Fuqua also said that this treatment could help fill in some of the gaps, by reaching people who can’t get a vaccine. “Vaccines rely on your immune system and so if you are immunocompromised, if you are elderly, if you have issues where you’re immune system doesn’t emit the appropriate response, if you’re allergic to the components of the vaccine, or things like that this is something that can work for you that doesn’t rely on your immune system for the response. It is just completely drug and virus interaction.”
The timeline for this treatment is still uncertain, so Fuqua doesn’t know when it might be available for the public if it is shown to be safe and effective in the clinical trials. The funding from the DoD will allow for the first phase of the clinical trials to take place in 2021 and Fuqua says they will apply for approval from the Food and Drug Administration once they get to that point.
File Photo //The Louisville Cardinal